Lifestyle
Reclassification of marijuana opens doors for much‑needed medical research into the benefits and risks of the drug
The DOJ’s move to reclassify medical marijuana as Schedule III could unlock long-blocked cannabis research—while raising new questions about safety, regulation, and risk.

Carey S. Cadieux, Binghamton University, State University of New York
When the U.S. Department of Justice moved to reclassify medical marijuana to a Schedule III drug on April 23, 2026, it set the stage for a vast amount of medical research that has been hobbled for decades by its more restrictive Schedule I classification.
The Justice Department also called for an expedited federal rescheduling process, with proceedings expected to begin in late June 2026, but for now cannabis at the federal level remains a Schedule I drug.
I’m an associate professor of nursing and I edited a textbook for nurses about providing care with cannabis. Cannabis is the umbrella term for the plant genus that includes both marijuana and hemp – two varieties of the same plant distinguished primarily by their content of THC, one of the active components of cannabis.
Moving cannabis to a Schedule III drug ushers in the end of the cannabis prohibition era and the beginning of the regulation era, potentially creating promising opportunities around research and new therapeutics.
How are drugs regulated by ‘schedule’?
The Controlled Substances Act of 1970 categorizes all substances regulated under existing federal law into one of five schedules. The act regulates the manufacturing, importation, possession, use and distribution of substances on each schedule.
Several factors determine schedule placement, including the drug’s medical use, scientific evidence of its benefits and pharmacological effects, patterns and history of abuse, public health risk level, degree of physical or psychological addiction potential, and whether the drug can be used to make another controlled substance.
The Drug Enforcement Administration’s rescheduling of marijuana will move it from its current classification as a Schedule I drug, defined as having a high risk for abuse and no accepted medical use, to a Schedule III drug under the Controlled Substances Act. While still tightly regulated, Schedule III drugs are considered to have moderate to low risk for physical and psychological dependence and to have some medical benefits.
Other Schedule I drugs include heroin, psilocybin, LSD, peyote and MDMA, or ecstasy. These drugs cannot be dispensed or prescribed, with some exceptions. Current Schedule III drugs include ketamine, anabolic steroids, testosterone, products with less than 90 milligrams of codeine per dosage unit and some cannabinoids.
The move to reclassify medical marijuana products as Schedule III drugs applies only to those products certified by state-level medical cannabis programs. All other cannabis products remain a federal Schedule I drug, including those available from states’ recreational cannabis programs.
Impacts of cannabis reclassification
This legal order acknowledges that medical marijuana has some medical value and asserts that it has a lower potential for abuse than under the previous Schedule I classification.
The reclassification also ensures that state-registered medical cannabis patients continue to be permitted to purchase medical cannabis products without changes to their current certification or recommendation.
One of the challenges with this new law is that states have not standardized medical cannabis regulations, and each state will have its own quality and testing standards. In Maine, for instance, medical cannabis is not tested for molds, fungus, heavy metals or pesticides, while recreational cannabis is.
This means that the Schedule III medical cannabis in Maine could be contaminated, while the state’s testing of recreational cannabis makes it much safer to consume.
What are the implications for marijuana research?
For decades, researchers have struggled to conduct high-quality research studies due to their lack of access to the cannabis products that patients actually use and restrictions on their processes.
With the reclassification, researchers who are registered with the DEA to research cannabis will be able to obtain cannabis flower and plant material, as well as manufactured cannabis products, such as tinctures and edibles, directly from state-licensed businesses that are DEA-registered.
This means researchers will no longer need to rely on the federal DEA registry for access to cannabis products for research, which were often inferior in quality and variety in comparison to the everyday products medical cannabis patients typically have access to. Instead, they will be able to study cannabis products that patients use in daily life, such as vapes and various edible products.
This shift in access will now allow researchers to undertake the gold standard of research approaches: the randomized controlled trial.
Randomized controlled trials will help researchers like my colleagues determine how effective cannabis is in treating people with complex medical needs. This includes patients who experience nausea and pain while undergoing cancer treatments, multiple sclerosis patients with severe muscle spasm and stiffness, and chronic pain patients who strive to find relief without using opioids.
Might rescheduling send mixed signals?
Rescheduling may lead people to believe that cannabis is safe for all people to consume.
However, a growing body of research points to possible adverse effects from cannabis use, particularly in vulnerable groups, such as people who are pregnant, adolescents, people with preexisting mental health conditions such as schizophrenia or psychosis, and those with cardiac issues.
Cannabis can also lead to adverse drug interactions. Therefore, medical patients should use it with discretion and under the guidance of a healthcare professional.
For most medical cannabis patients, THC doses should start low and gradually be increased.
Rescheduling will be a big step toward helping researchers build a greatly needed solid body of evidence around both the benefits and potential harms of cannabis. But rescheduling should not be interpreted as a signal that cannabis is harmless.
Carey S. Cadieux, Associate Professor of Nursing, Binghamton University, State University of New York
This article is republished from The Conversation under a Creative Commons license. Read the original article.
Our Lifestyle section on STM Daily News is a hub of inspiration and practical information, offering a range of articles that touch on various aspects of daily life. From tips on family finances to guides for maintaining health and wellness, we strive to empower our readers with knowledge and resources to enhance their lifestyles. Whether you’re seeking outdoor activity ideas, fashion trends, or travel recommendations, our lifestyle section has got you covered. Visit us today at https://stmdailynews.com/category/lifestyle/ and embark on a journey of discovery and self-improvement.
Health
The War on Sunscreen: How Misinformation May Be Undermining Cancer Prevention
Despite its wide recognition as an effective tool for skin cancer prevention, many Americans still say they aren’t sure how sunscreen works. This sunscreen confusion highlights a central challenge in preventing skin cancer, which is the most common form of cancer in the United States.

(Feature Impact) Despite its wide recognition as an effective tool for skin cancer prevention, many Americans still say they aren’t sure how sunscreen works. In fact, confusion about its use and misinformation persist, especially on social media.
According to a national survey conducted by Atomik Research on behalf of the nonprofit Melanoma Research Alliance (MRA), 53% of the 2,000 adult respondents said they’ve seen claims that sunscreen ingredients may be harmful while 59% reported they’re concerned about what’s in sunscreen and 38% don’t believe sunscreen is safe and effective.
This sunscreen confusion highlights a central challenge in preventing skin cancer, which is the most common form of cancer in the United States, according to the U.S. Centers for Disease Control and Prevention.
 Among the various types of skin cancer, 9 out of 10, including melanoma, are linked to exposure to ultraviolet (UV) radiation, according to the MRA. The survey found most Americans have a basic understanding of the risks of sun exposure, including the more than 8 in 10 who recognize spending long hours in the sun contributes to melanoma risk. However, roughly one-quarter said they rarely or never use sunscreen when outdoors.
Among the various types of skin cancer, 9 out of 10, including melanoma, are linked to exposure to ultraviolet (UV) radiation, according to the MRA. The survey found most Americans have a basic understanding of the risks of sun exposure, including the more than 8 in 10 who recognize spending long hours in the sun contributes to melanoma risk. However, roughly one-quarter said they rarely or never use sunscreen when outdoors.
Reducing exposure to UV radiation lowers the risk of skin cancer, making sunscreen a key part of prevention, even during those everyday moments you may not immediately recognize as “risky,” like a short walk through a parking lot or an hour sitting in the bleachers at a sporting event.
To combat the confusion, it’s important to first understand how sunscreen works. It absorbs or blocks UV radiation from reaching the skin, preventing DNA damage that can cause cancer.
“Conflicting or incomplete information can make people less likely to wear sunscreen consistently,” said Dr. Joan Levy, chief science officer at the MRA, “but the science on sun protection is clear, and sunscreen is one of the most effective tools to prevent melanoma.”
Active ingredients in sunscreen in the U.S. undergo rigorous review by the Food and Drug Administration (FDA), which evaluates them as over-the-counter drugs – a standard which requires extensive testing and contributes to a more limited set of approved UV filters compared to Europe, where sunscreens are regulated as cosmetics.
The FDA is evaluating additional methodologies for assessing sunscreen ingredients, a process that could, according to Levy, expand the number of approved UV filters available in the U.S.
While melanoma remains the deadliest form of skin cancer, real progress is being made in melanoma research. Recent advances are improving outcomes for many patients with the disease, though approximately 50% of patients do not respond to current treatments, according to the MRA, underscoring why prevention and early detection are critical.
“Treatments such as immunotherapy have significantly improved outcomes for many melanoma patients, but we should never need to treat what we could have prevented,” Levy said.
To learn more and find strategies to protect your skin, visit CureMelanoma.org.
Photo courtesy of Shutterstock


SOURCE:
Our Lifestyle section on STM Daily News is a hub of inspiration and practical information, offering a range of articles that touch on various aspects of daily life. From tips on family finances to guides for maintaining health and wellness, we strive to empower our readers with knowledge and resources to enhance their lifestyles. Whether you’re seeking outdoor activity ideas, fashion trends, or travel recommendations, our lifestyle section has got you covered. Visit us today at https://stmdailynews.com/category/lifestyle/ and embark on a journey of discovery and self-improvement.
Child Health
9 Steps for a Lifetime of Heart-Healthy Eating
Heart Healthy: More than half of adults and about 60% of children in the United States have unhealthy diets, which can contribute to higher rates of health factors such as high blood pressure and obesity. However, these nine key features of a heart-healthy dietary pattern may help reduce cardiovascular disease risk and improve quality of life.

9 Steps for a Lifetime of Heart-Healthy Eating
(Feature Impact) More than half of adults and about 60% of children in the United States have unhealthy diets, which can contribute to higher rates of health factors such as high blood pressure and obesity, and lead directly to poor health outcomes including death from chronic conditions such as cardiovascular disease.
These findings come from the American Heart Association, a global force changing the future of health for all, which published its “2026 Dietary Guidance to Improve Cardiovascular Health” in its peer-reviewed, flagship journal “Circulation” recommending Americans follow a lifelong healthy eating pattern that may reduce cardiovascular disease risk and improve quality of life based on the latest in evidence-based science.
The statement outlines nine key features of a heart-healthy dietary pattern:
- Adjust energy intake and expenditure to achieve and maintain a healthy body weight: Try to balance how much you eat with how active you are to reach and maintain a healthy body weight.
- Eat a variety of vegetables and fruits: Include different colors, textures and types of produce. Remember, even canned and frozen can be nutritious and affordable.
- Choose foods made mostly with whole grains: Foods such as whole‑wheat bread, brown rice and oatmeal are typically better choices than refined grains including white bread or rice.
- Choose healthy sources of protein: Shift from meat to plant-based sources such as legumes, including beans, peas and lentils, along with nuts and seeds; regularly consume fish and seafood; select low-fat or fat-free dairy products; and, if red meat is desired, choose lean cuts, avoid processed forms and limit portion size.
- Choose sources of unsaturated fats: Replace saturated fats with healthy unsaturated fats, including those from nuts, seeds, avocados and nontropical plant oils.
- Choose minimally processed foods: Go with foods close to their natural state, with minimal added commercial ingredients, rather than those that are highly processed with additives.
- Minimize intake of added sugars: Limit the sugar-sweetened beverages you drink and the foods with added sugar you eat.
- Choose foods low in sodium and prepare foods with minimal or no salt: Be aware of hidden sources of sodium in prepared and packaged foods, and season food with healthier options such as herbs, spices or lemon instead of salt.
- If alcohol is not consumed, do not start; if alcohol is consumed, limit intake: Alcohol can increase your risk for high blood pressure and other health conditions, so if you don’t drink, don’t start.
“For healthy eating to be more attainable and sustainable, we recommend people focus on their overall eating pattern rather than specific nutrients or foods,” said Alice H. Lichtenstein, D.Sc., FAHA, volunteer chair of the scientific statement writing committee and senior scientist and leader of the Diet and Chronic Disease Prevention Directive at the Jean Mayer USDA Human Nutrition Research Center on Aging at Tufts University. “Every time you choose to make a swap for a healthier alternative, you’re making a step toward a healthier life.”
While the updated guidance is specifically designed to improve cardiovascular health, it’s generally consistent with dietary recommendations for other conditions like Type 2 diabetes, kidney disease, some cancers and brain health as well due to shared risk factors, including high blood pressure, high cholesterol, high blood sugar, excess weight and reduced kidney function.
“A healthy dietary pattern can support lifelong health and well-being beyond cardiovascular health,” Lichtenstein said.
To learn more about eating for heart and brain health, visit heart.org/healthydietand talk to your health care provider about what will work best for you.
Photo courtesy of Shutterstock


SOURCE:
The Food and Drink section at STM Daily News delivers fresh coverage on dining, drinks, recipes, and the food stories bringing communities together.
Lifestyle
You have 30 minutes to save a knocked-out tooth. Only 27% of U.S. adults know that.
Saving a knocked-out tooth: Most U.S. adults say they would do almost anything to avoid losing a natural tooth. Most have also put off going to the dentist when they suspected something was wrong. And some may not be aware of the dental specialists whose entire focus is helping them do exactly that.

You have 30 minutes to save a knocked-out tooth. Only 27% of U.S. adults know that.
(Tiffany Miller) Most U.S. adults say they would do almost anything to avoid losing a natural tooth. Most have also put off going to the dentist when they suspected something was wrong. And some may not be aware of the dental specialists whose entire focus is helping them do exactly that. A new national survey from the American Association of Endodontists, the organization representing those specialists, finds all three of those things together at the center of how U.S. adults actually approach their dental health.
That tension runs through the entire survey. When faced with a serious tooth infection, 61% would choose a treatment that saves the tooth over extraction. Seventy-eight percent say they would do almost anything to avoid losing a natural tooth. The attachment runs deeper than appearances. It is about the tooth itself, the one that has been there your whole life, that feels irreplaceable in a way an implant or crown never quite does.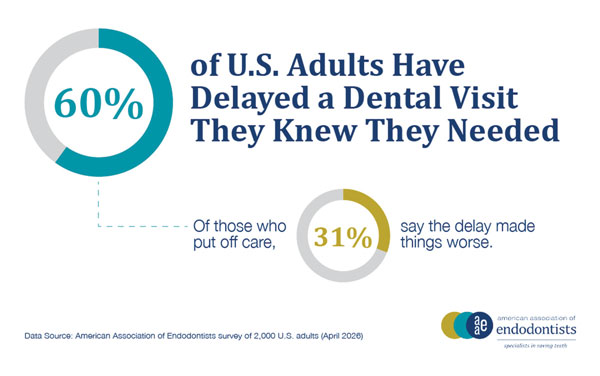
The gap is not in conviction. It is in knowing what to do.
Only 27% of U.S. adults correctly identify that a knocked-out tooth needs to be treated within 30 minutes to give it the best chance of being saved. The rest either guess wrong or have no idea how quickly the clock is running. And the specialists most equipped to help in that moment, dental professionals called endodontists whose entire focus is saving natural teeth, are known to only 44% of U.S. adults.
Cost widens the gap. Sixty percent of U.S. adults admit to having put off going to the dentist when they suspected something was wrong. The anatomy of that delay is familiar: a symptom that does not seem urgent enough, a bill that feels easier to avoid than face, an appointment that keeps getting pushed to next month. Of those who waited, 51% say it made things worse.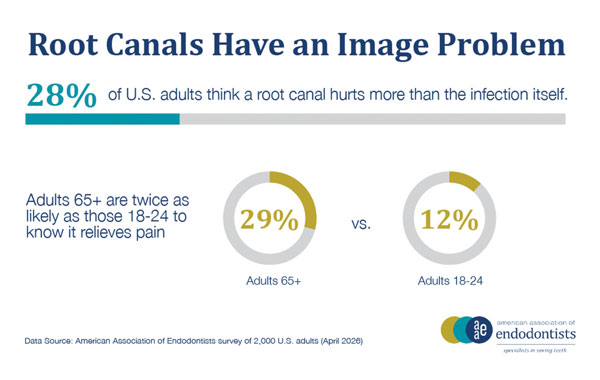
And then there is the fear. Root canals have a reputation that the data does not support. Asked what they believe, 28% of U.S. adults say a root canal is usually more painful than the toothache that sent them to the dentist in the first place. They have it backward. A root canal is designed to relieve that pain, not add to it, according to the American Association of Endodontists. But the perception lingers, shaped in part by a fragmented information environment where 35% of U.S. adults say they have encountered conflicting or confusing information online about procedures such as root canals.
U.S. adults feel strongly about their natural teeth, probably more than they would have predicted before being asked. They delay the care that protects them, do not know what to do when time is short, and are not always getting their information from sources that serve them well.
For a tooth that can often be saved in 30 minutes, that combination has consequences.
Methodology
American Association of Endodontists commissioned Atomik Research, a creative market research agency part of 4media group, to conduct an online survey of 2,000 adults throughout the United States. The margin of error is plus or minus 2 percentage points at a 95 percent confidence level. Fieldwork was conducted between April 3 and April 8, 2026.
Photo courtesy of Shutterstock


SOURCE:
American Association of Endodontists
Stay informed with the stories shaping tomorrow at STM Daily News — your source for breaking news, transportation, technology, UAP updates, science, entertainment, and more. Join our growing community and subscribe to the STM Daily News newsletter for curated headlines, exclusive updates, and featured stories delivered directly to your inbox. Don’t miss what’s next.
