
FDA CDC News
Nationwide Artificial Tears Lubricant Eye Drops Recall Due to Possible Contamination
Last Updated on February 2, 2023 by Daily News Staff
Global Pharma Healthcare is voluntarily recalling all lots within expiry of their Artificial Tears Lubricant Eye Drops, distributed by /EzriCare, LLC- and Delsam Pharma, to the consumer level, due to possible contamination. The Centers for Disease Control and Prevention (CDC) alerted FDA to an investigation of a multi-state cluster of Verona Integron-mediated Metallo-β-lactamase (VIM)- and Guiana-Extended Spectrum-β-Lactamase (GES)- producing carbapenem-resistant Pseudomonas aeruginosa (VIM-GES-CRPA) infections possibly associated with the use of the artificial tears manufactured by Global Pharma Healthcare. To date, there are 55 reports of adverse events including eye infections, permanent loss of vision, and a death with a bloodstream infection.
Risk Statement: Use of contaminated artificial tears can result in the risk of eye infections that could result in blindness.
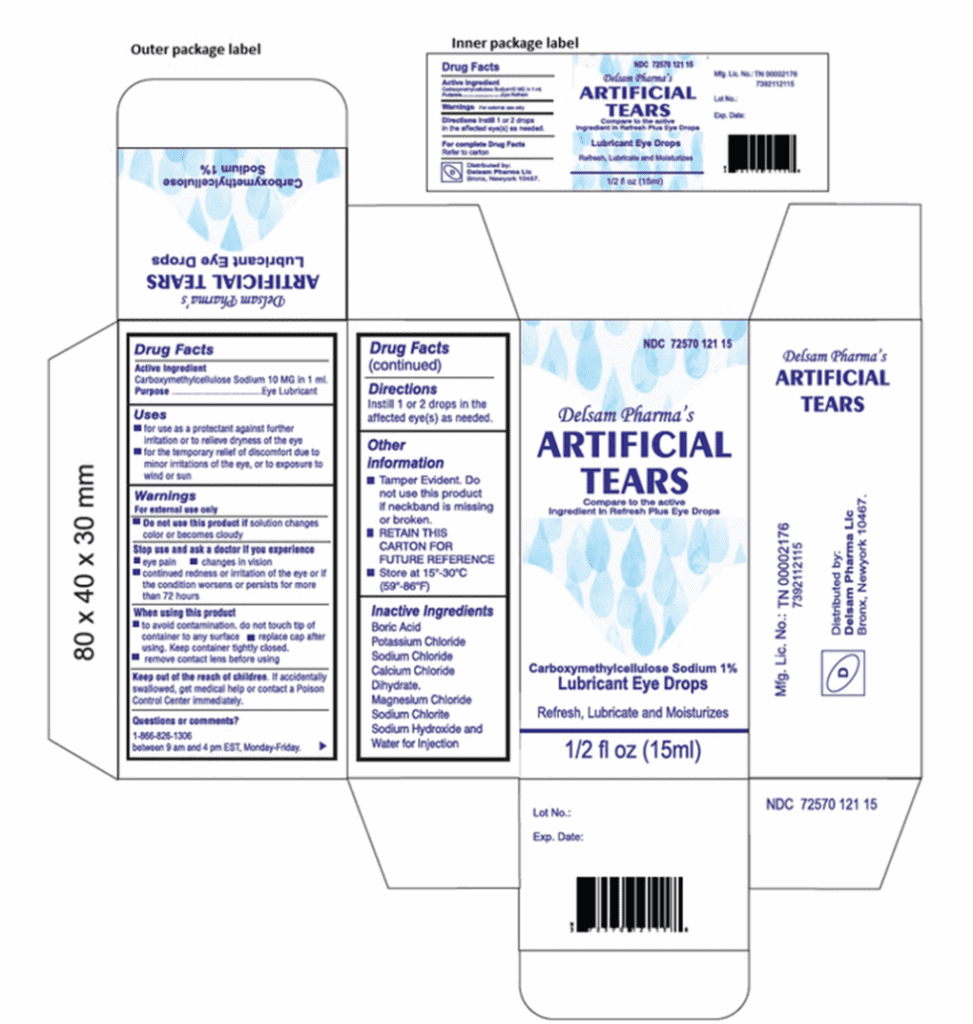
Artificial Tears (carboxymethylcellulose sodium) Lubricant Eye Drops, 10 mg in 1 mL, ½ fl oz (15 ml) bottle are used as a protectant against further irritation or to relieve dryness of the eye for the temporary relief of discomfort due to minor irritations of the eye, or to exposure to wind or sun. The product is packaged in a bottle with a safety seal and are placed in a carton box Ezricare NDC 79503-0101-15, UPC 3 79503 10115 7; Delsam Pharma’s NDC 72570-121-15, UPC -72570-0121-15. It can be identified by the photos below. The product was distributed Nationwide in the USA over the Internet.
Global Pharma Healthcare is notifying the distributors of this product, Aru Pharma Inc. and Delsam Pharma and is requesting that wholesalers, retailers and customers who have the recalled product should stop use.
Consumers with questions regarding this recall can contact the distributors: Aru Pharma/Ezricare, LLC – by phone: 1-518-738-7602 or by e-mail: arupharmainc@yahoo.com from Monday to Friday, 11am to 4pm EST; or DELSAM Pharma LLC by phone: 1-866-826-1309 or by e-mail: delsampharma@yahoo.com from Monday to Friday from 11am to 4pm EST. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to using these over-the-counter drug products.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online
- Regular Mail or Fax: Download form or call 1- 800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178

This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
Source: FDA
Discover more from Daily News
Subscribe to get the latest posts sent to your email.
Food and Beverage
Diva Fam Inc. Announces Voluntary Recall of True Sea Moss “Sea Moss Gel Superfood” Products Due to Possible Health Risk
Diva Fam Inc. is recalling all True Sea Moss Sea Moss Gel Superfood flavors nationwide due to missing pH/temperature records and potential botulism risk.

Diva Fam Inc.. announced a voluntary recall of all lots and flavors of its True Sea Moss brand Sea Moss Gel Superfood due to a lack of required regulatory authorization and temperature monitoring records for pH-controlled food products, according to a company statement released January 9, 2026.
The company said the recall applies to products manufactured prior to January 9, 2026. The manufacture date (MFD) is indicated on the can lid in MM/YYYY format.
Why the products are being recalled
Diva Fam said the recall is related to missing required regulatory authorization and temperature monitoring records for certain pH-controlled food products. The company noted that pH-controlled foods that are not manufactured in accordance with applicable regulatory requirements may present a potential risk of microbial growth, including organisms that can produce toxins associated with botulism.

Botulism is a rare but serious illness that can affect the nervous system. Symptoms may include general weakness, dizziness, double vision, difficulty speaking or swallowing, and, in severe cases, difficulty breathing or muscle weakness.
Diva Fam said no illnesses or adverse health events have been reported in connection with the products subject to this recall to date.
Where the products were sold
The affected products were distributed nationwide through select retail locations, online via https://truеsеamоss.cоm/, and other distribution channels, according to the company.
Recalled products (all flavors, all lots)
The recall includes all flavors and sizes and batch numbers of True Sea Moss brand Sea Moss Gel Superfood packaged in 16 FL OZ (473 mL) glass jars, manufactured prior to January 9, 2026.
Recalled flavors and UPCs
| Flavor | UPC |
|---|---|
| Mango | 5065006235875 |
| Pineapple | 5065006235288 |
| Wildcrafted | 5065006235073 |
| Apple and Cinnamon | 5065006235776 |
| Elderberry | 5065006235189 |
| Passion Fruit | 5061033691882 |
| Blue Spirulina and Raspberry | 5065006235813 |
| Strawberry | 5065006235271 |
| Cherry | 5061033691264 |
| Mango and Pineapple | 5065006235301 |
| 5 Blends in 1 | 5061033690052 |
| Soursop | 5061033691875 |
| Lemon Pie | 5061033691271 |
| Orange | 5061033692926 |
How the issue was identified
The company said the matter was identified during a California Department of Public Health inspection that raised questions regarding regulatory authorization and related production records for certain distributed products. Diva Fam said it is cooperating fully with regulatory authorities and initiated the voluntary recall to ensure regulatory alignment.
The company said the recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
What consumers should do
- Discontinue use of the affected product.
- Follow the instructions provided by the place of purchase regarding product return or disposal.
- Contact the company for additional information (details below).
Consumer and media contact
Consumers seeking additional information may contact:
- Email: support@divafam.com
- Phone: (818) 751-3882
- Hours: Monday through Friday, 9:00 a.m. – 5:00 p.m. Pacific Time
Source: Diva Fam Inc. (PRNewswire, Jan. 9, 2026)
https://stmdailynews.com/culvers-thank-you-farmers-project-hits-8-million-donation-milestone/
Discover more from Daily News
Subscribe to get the latest posts sent to your email.
Women's Health
Is Hormone Replacement Therapy Safe? What the FDA’s New Decision Means for Menopause Treatment
For more than 20 years, hormone replacement therapy for menopause has carried a warning label from the Food and Drug Administration describing the medication’s risk of serious harms – namely, cancer, cardiovascular disease and possibly dementia.
Last Updated on November 19, 2025 by Daily News Staff

Our Lifestyle section on STM Daily News is a hub of inspiration and practical information, offering a range of articles that touch on various aspects of daily life. From tips on family finances to guides for maintaining health and wellness, we strive to empower our readers with knowledge and resources to enhance their lifestyles. Whether you’re seeking outdoor activity ideas, fashion trends, or travel recommendations, our lifestyle section has got you covered. Visit us today at https://stmdailynews.com/category/lifestyle/ and embark on a journey of discovery and self-improvement.
Discover more from Daily News
Subscribe to get the latest posts sent to your email.
News
Nationwide Shrimp Recall Expands to Arizona: What You Need to Know
Nationwide Shrimp Recall: AquaStar has recalled Kroger, Kroger Mercado, and AquaStar frozen shrimp in Arizona and other states due to possible cesium-137 contamination. Check UPCs, lot codes, and best-by dates to see if your shrimp is affected.
Last Updated on September 26, 2025 by Daily News Staff
Steamed shrimp on plate
Nationwide Shrimp Recall Expands to Arizona: What You Need to Know
A major frozen shrimp recall is currently underway across the United States — and Arizona shoppers are directly affected. AquaStar (USA) Corp has announced a recall of multiple frozen shrimp products, both raw and cooked, due to potential contamination with cesium-137 (Cs-137), a radioactive substance.
Products Included in the Recall
The recall covers several popular brands and package types, including:
Kroger Raw Colossal EZ Peel Shrimp (2 https://stmdailynews.com/cash-trapping-how-to-protect-yourself-from-this-sneaky-atm-scam/ bag)
Kroger Mercado Cooked Medium Peeled Tail-Off Shrimp (2 lb bag)
AquaStar Raw Peeled Tail-On Shrimp Skewers (1.25 lb bag)
AquaStar Cocktail Shrimp trays (sold at Walmart and other retailers)
In total, more than 85,000 packages of shrimp have been pulled from stores nationwide. These products were distributed to several states, including Arizona, between June and September 2025.
Why the Recall?
Routine testing detected the presence of cesium-137, a radioactive contaminant. While no illnesses or adverse reactions have been reported, long-term exposure to Cs-137 may increase the risk of certain cancers. Out of caution, the FDA and AquaStar urge consumers not to eat these shrimp.
🔍 How to Identify the Recalled Shrimp
Shoppers should look at UPC codes, lot codes, and best-by dates printed on the packaging. Here are the specific products under recall:
Product | UPC | Lot Code(s) | Best-By Date(s) |
|---|---|---|---|
Kroger Raw Colossal EZ Peel Shrimp (2 lb) | 20011110643906 | 10662 5085 10 · 10662 5097 11 · 10662 5106 11 · 10662 5107 10 · 10662 5111 11 · 10662 5112 10 · 10662 5113 10/11 · 10662 5114 10/11 | March 26 2027 · April 7 2027 · April 16–24 2027 |
Kroger Mercado Cooked Medium Peeled Tail-Off Shrimp (2 lb) | 011110626196 | 10662 5112 11 · 10662 5113 10 | October 22–23 2027 |
AquaStar Raw Peeled Tail-On Shrimp Skewers (1.25 lb) | 731149390010 | 10662 5127 10 · 10662 5128 11 · 10662 5133 11 · 10662 5135 10 | November 7–15 2027 |
AquaStar Cocktail Shrimp Trays (Walmart) | 19434612191 | 10662 5106 · 10662 5107 · 10662 5124 · 10662 5125 | Dates vary by lot |
What Should Consumers Do?
Check your freezer for the affected shrimp products.
Do not eat them. If you have the recalled shrimp, throw it away or return it to the store where it was purchased.
Stay updated. The FDA continues to monitor the situation and will provide further updates as needed.
No Reported Illnesses So Far
Although the recall sounds alarming, health officials stress that no illnesses have been linked to these shrimp products at this time. The move is a precaution to protect consumers.
👉 Bottom line for Arizona shoppers: If you’ve bought frozen shrimp from Kroger, Kroger Mercado, or AquaStar between June and September 2025, check the packaging details immediately. When in doubt, don’t eat it.
🔗 Resources for More Information
FDA Recall Notice – AquaStar USA Corp Recalls Kroger & AquaStar Frozen Shrimp
FDA Advisory – Do Not Eat, Sell, or Serve Certain Imported Frozen Shrimp
STM Daily News
Cash Trapping: How to Protect Yourself from This Sneaky ATM ScamLink: https://stmdailynews.com/cash-trapping-how-to-protect-yourself-from-this-sneaky-atm-scam/
Discover more from Daily News
Subscribe to get the latest posts sent to your email.
